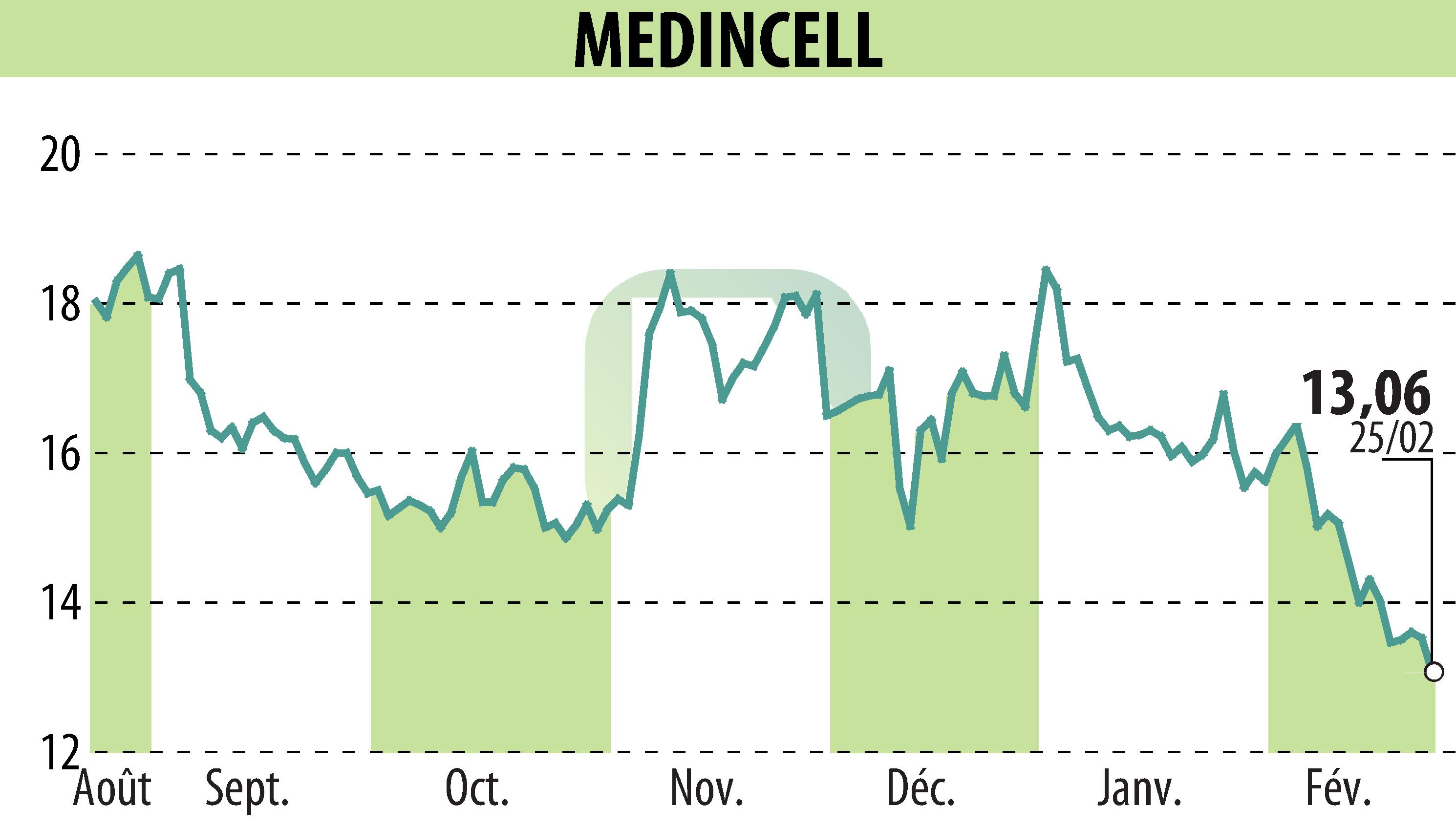
on MEDINCELL (EPA:MEDCL)
FDA Accepts Application for UZEDY's Use in Treating Bipolar I Disorder
Teva Pharmaceuticals and Medincell have announced that the FDA has accepted their application for expanding the indication of UZEDY®, an antipsychotic injectable, to include treatment for Bipolar I Disorder (BP-I). UZEDY is already approved in the U.S. for treating schizophrenia in adults via a once-monthly or bi-monthly injection.
The new application is based on existing clinical data of UZEDY and FDA conclusions on previously approved risperidone formulations for BP-I. This acceptance underscores Teva's commitment to advancing neuroscience treatments.
UZEDY's long-acting injectable form addresses unmet needs in BP-I treatment, potentially improving patient adherence. Teva will oversee regulatory processes and commercialization, with Medincell eligible for royalties on net sales.
R. H.
Copyright © 2025 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all MEDINCELL news
