on Heidelberg Pharma AG (isin : DE000A11QVV0)
Heidelberg Pharma Advances HDP-101 in Phase I/IIa Study
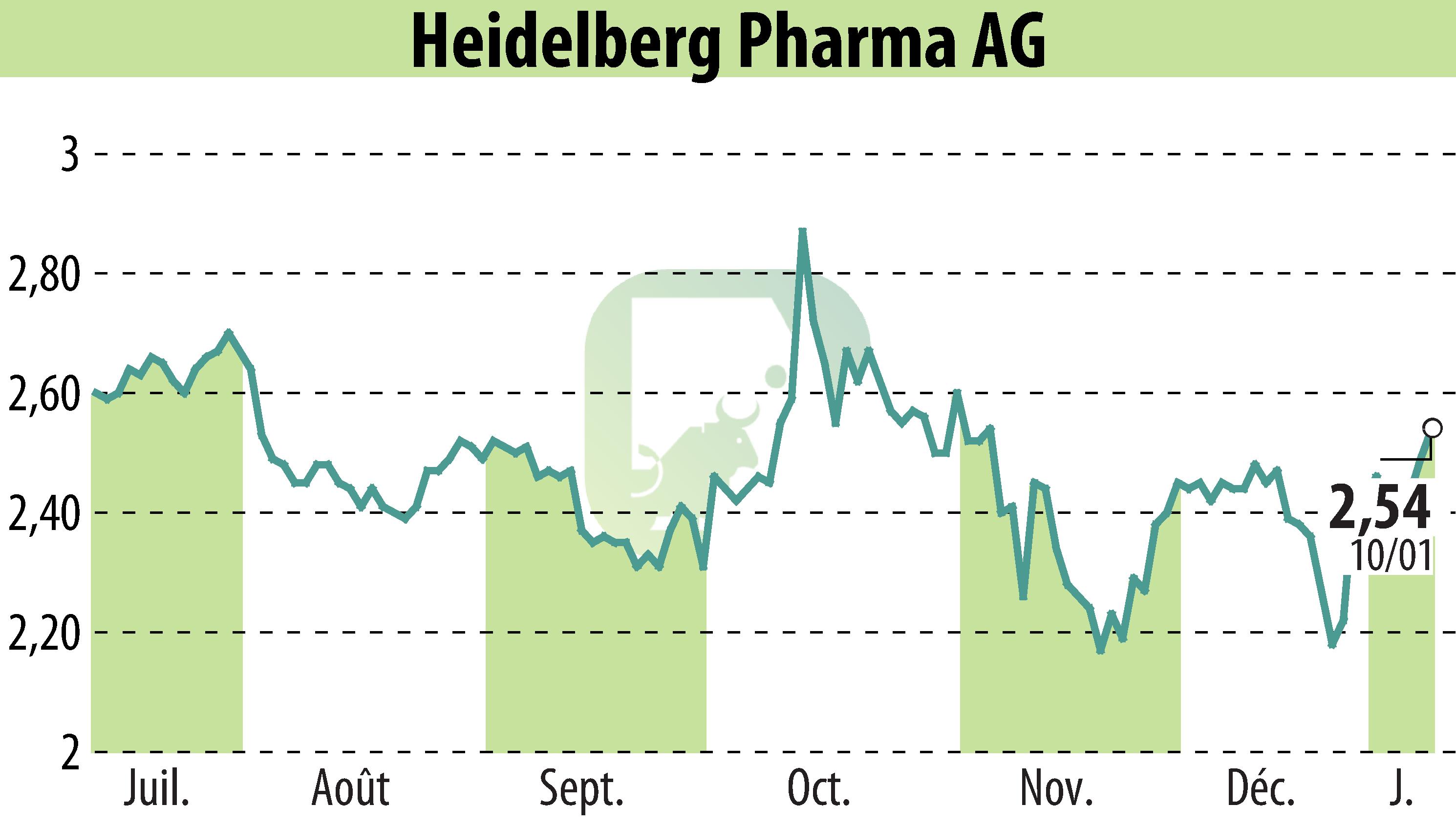
Heidelberg Pharma AG announced the progression into Cohort 7 in their Phase I/IIa study of HDP-101, an ATAC candidate targeting multiple myeloma. HDP-101 has been well tolerated with no dose-limiting toxicities noted. A patient in Cohort 5 achieved complete remission, indicating potential efficacy. This advancement marks a significant step in developing new treatment options for relapsed or refractory multiple myeloma.
The study has successfully implemented dose optimization strategies in Cohort 6 with no toxicities across 10 patients, allowing the Safety Review Committee to recommend progression with an escalated dose in Cohort 7. This phase involves two parallel treatment arms, one with additional premedication to enhance safety.
The goal is to assess safety, tolerability, and efficacy as the study enrolls patients with BCMA-expressing plasma cell disorders. Promising results strengthen Heidelberg Pharma’s position in the oncology treatment landscape.
R. H.
Copyright © 2025 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all Heidelberg Pharma AG news
