on MIRA Pharmaceuticals (NASDAQ:MIRA)
MIRA Pharmaceuticals Advances with Phase 1 Trial for Ketamir-2
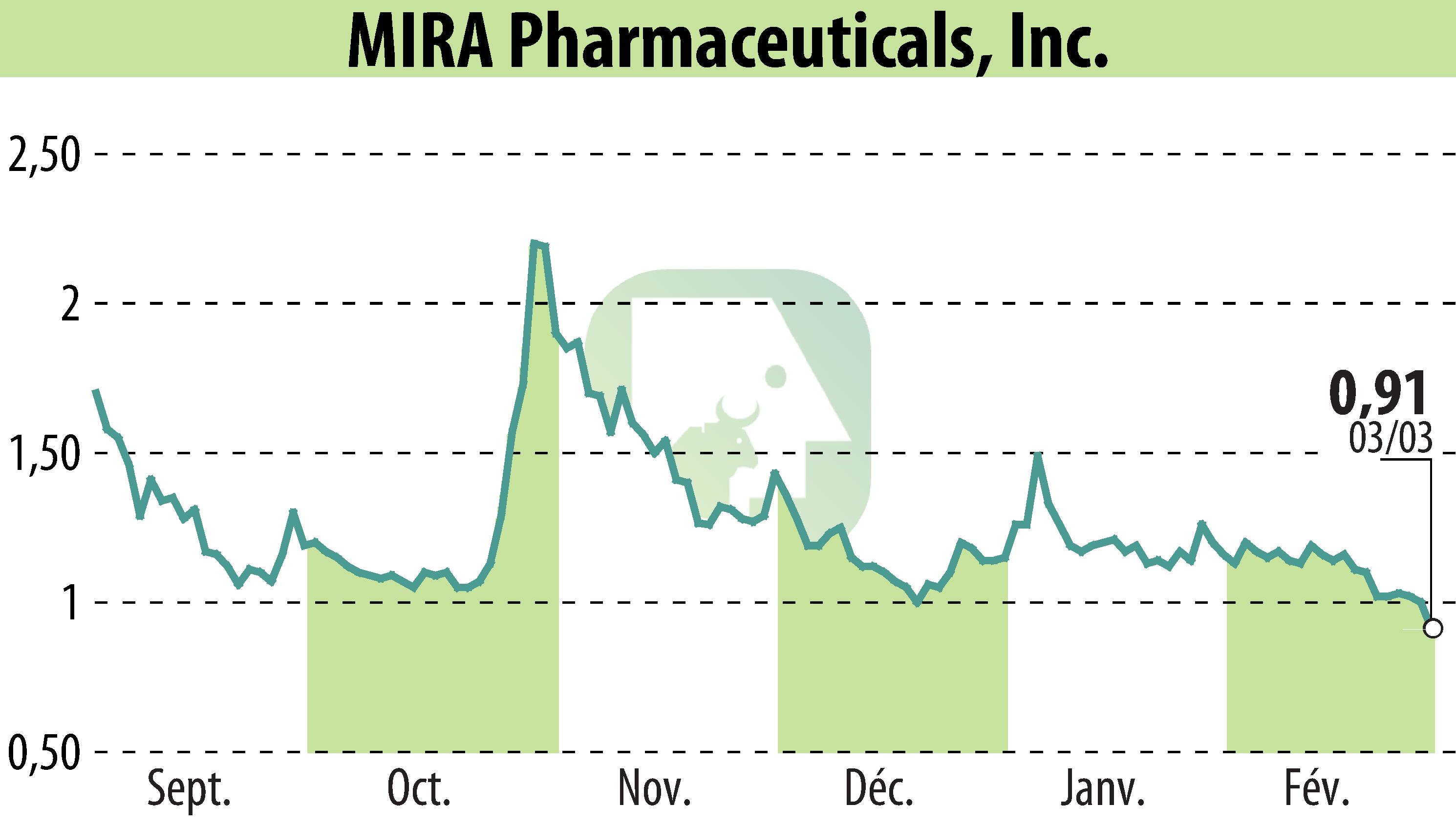
MIRA Pharmaceuticals has announced the approval and upcoming initiation of a Phase 1 clinical trial for their novel oral ketamine analog, Ketamir-2, intended for neuropathic pain. The trial, set in Jerusalem, will evaluate safety, tolerability, and pharmacokinetics in healthy adults, and is expected to conclude by Q4 2025.
The approval comes from the Israeli Ministry of Health. The study, involving 56 participants over two parts, will also assess ketamine-related side effects. By opting for Israel, MIRA seeks to optimize costs and timelines, facilitating an anticipated Phase 2a study starting later in the year.
Preclinical studies highlighted Ketamir-2's efficacy in reversing pain and improving safety over existing treatments like gabapentin and pregabalin. With an unmet market for neuropathic pain, Ketamir-2 shows promise as a safer alternative. MIRA is also exploring broader applications, including PTSD and localized pain relief.
R. H.
Copyright © 2025 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all MIRA Pharmaceuticals news
