on MIRA Pharmaceuticals (NASDAQ:MIRA)
MIRA Pharmaceuticals Submits IND for Ketamir-2 to FDA
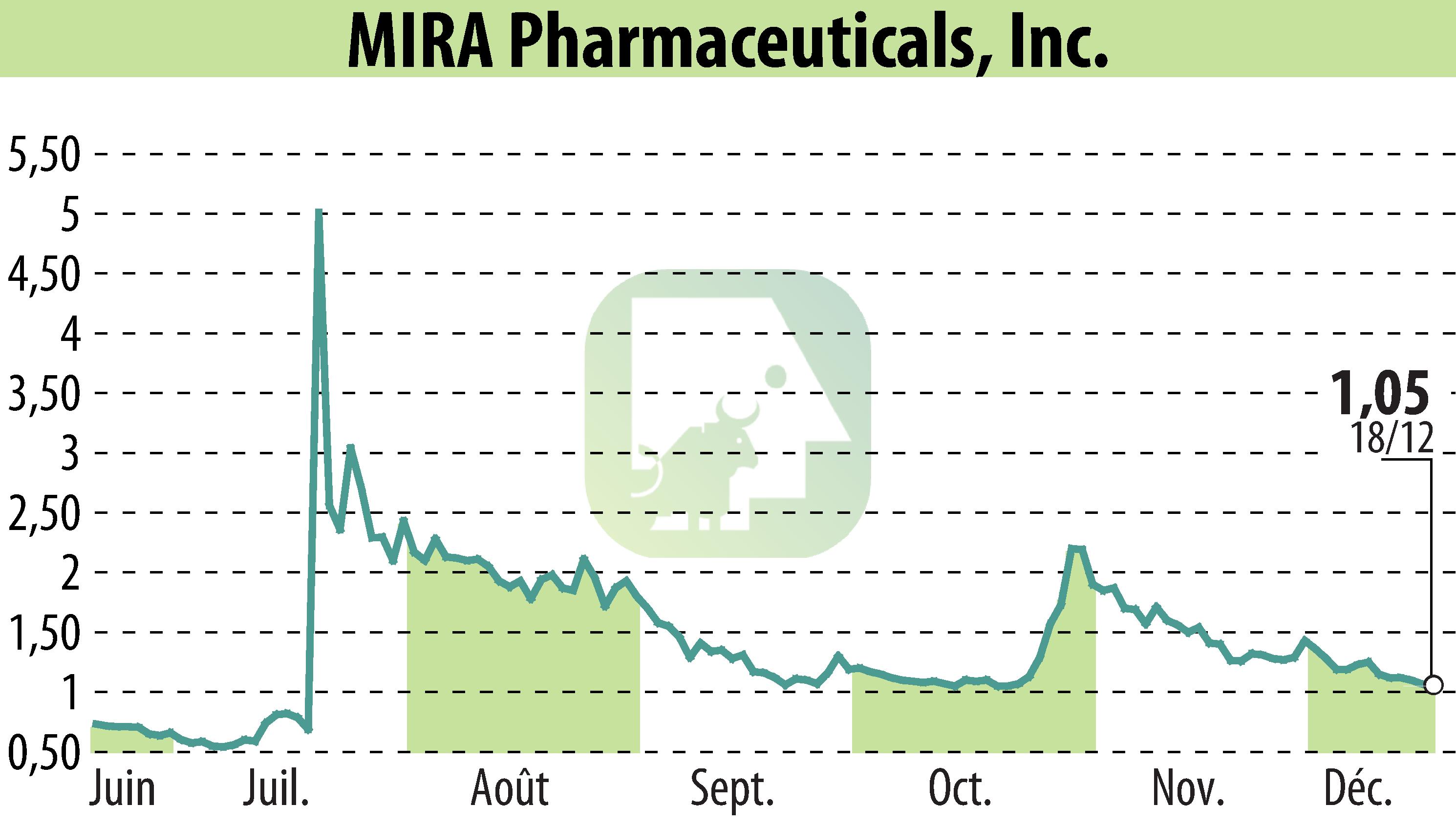
MIRA Pharmaceuticals, Inc. has submitted an Investigational New Drug (IND) application to the FDA for Ketamir-2. This marks a significant step for its potential treatment of neuropathic pain. The company aims to advance its lead drug candidate by beginning Phase I trials in early 2025.
Ketamir-2 is an oral ketamine analog targeting neuropathic pain and other neuropsychiatric conditions. It promises reduced side effects compared to existing treatments by selectively targeting the NMDA receptor. It is notably not classified as a controlled substance, simplifying regulatory hurdles.
The neuropathic pain market is projected to grow significantly by 2030. Current treatments like gabapentin and opioids have limitations, which Ketamir-2 aims to overcome. Upcoming clinical trials will assess its safety and efficacy.
R. P.
Copyright © 2025 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all MIRA Pharmaceuticals news
