on Moderna, Inc. (NASDAQ:MRNA)
Moderna Files FDA Application for JN.1 Targeting COVID-19 Vaccine
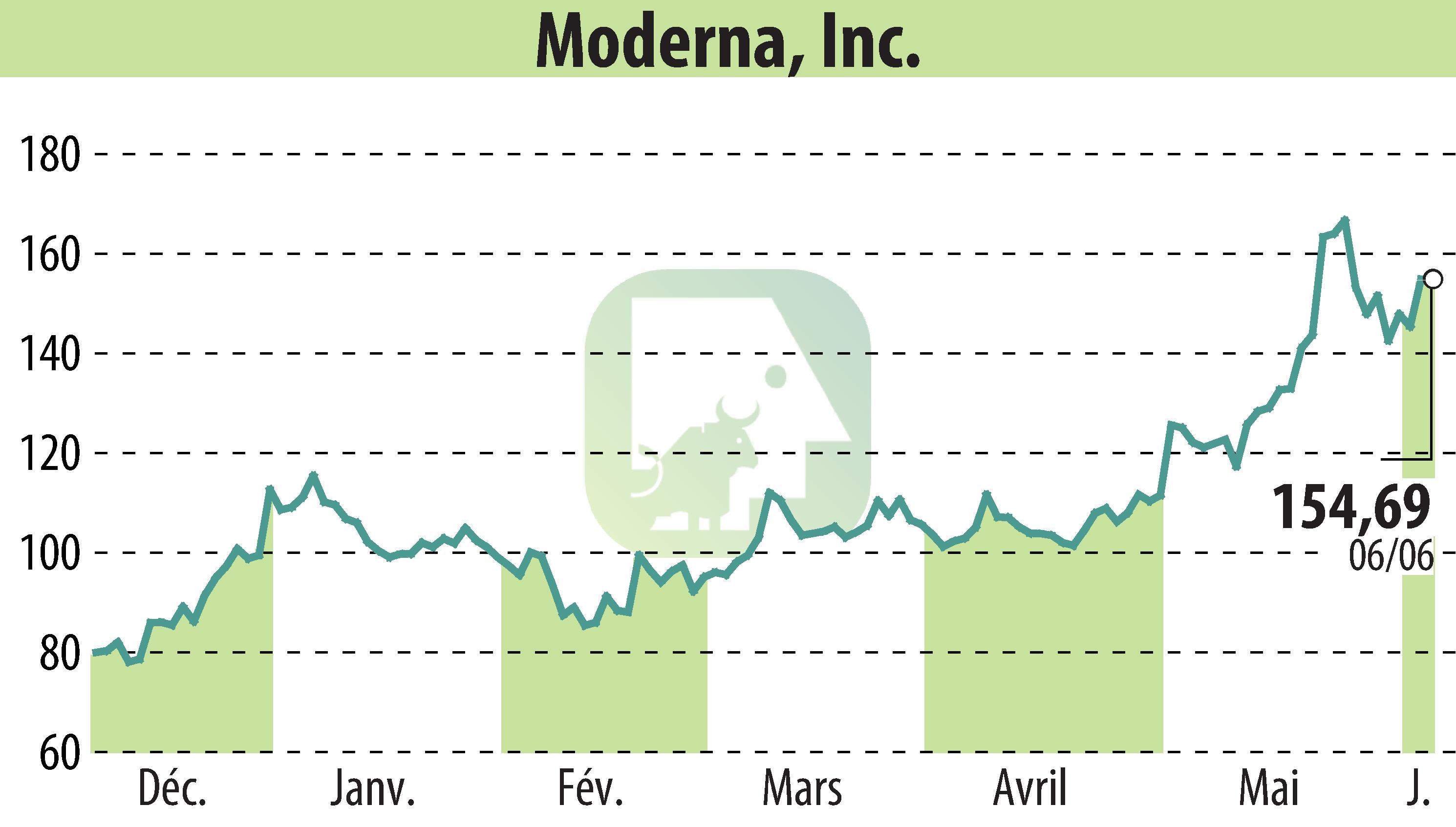
CAMBRIDGE, MA / ACCESSWIRE / June 7, 2024 / Moderna, Inc. (NASDAQ:MRNA) has taken a significant step in the fight against COVID-19. The company announced it has submitted an application to the U.S. Food and Drug Administration (FDA) for its Spikevax 2024-2025 formula. This new version of the vaccine targets the SARS-CoV-2 variant JN.1. Manufacturing is in progress, and doses will be ready for shipping as early as August, pending regulatory approval.
The submission follows FDA guidance that COVID-19 vaccines for the 2024-2025 season should be updated to a monovalent JN.1 composition. This aligns with recommendations from the World Health Organization (WHO) and the European Medicines Agency (EMA). Most solicited local adverse events reported include injection site pain, while systemic adverse events include headache, fatigue, myalgia, and chills.
Moderna’s CEO, Stéphane Bancel, emphasized the importance of staying current with COVID-19 vaccinations to protect against respiratory illnesses. The company is also submitting data to regulators worldwide to facilitate the registration and supply of its updated Spikevax formula in time for the next vaccination season.
R. H.
Copyright © 2026 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all Moderna, Inc. news
