on MONOGRAM ORTHOPAEDICS INC (NASDAQ:MGRM)
Monogram Technologies Secures FDA Clearance for mBôs TKA System
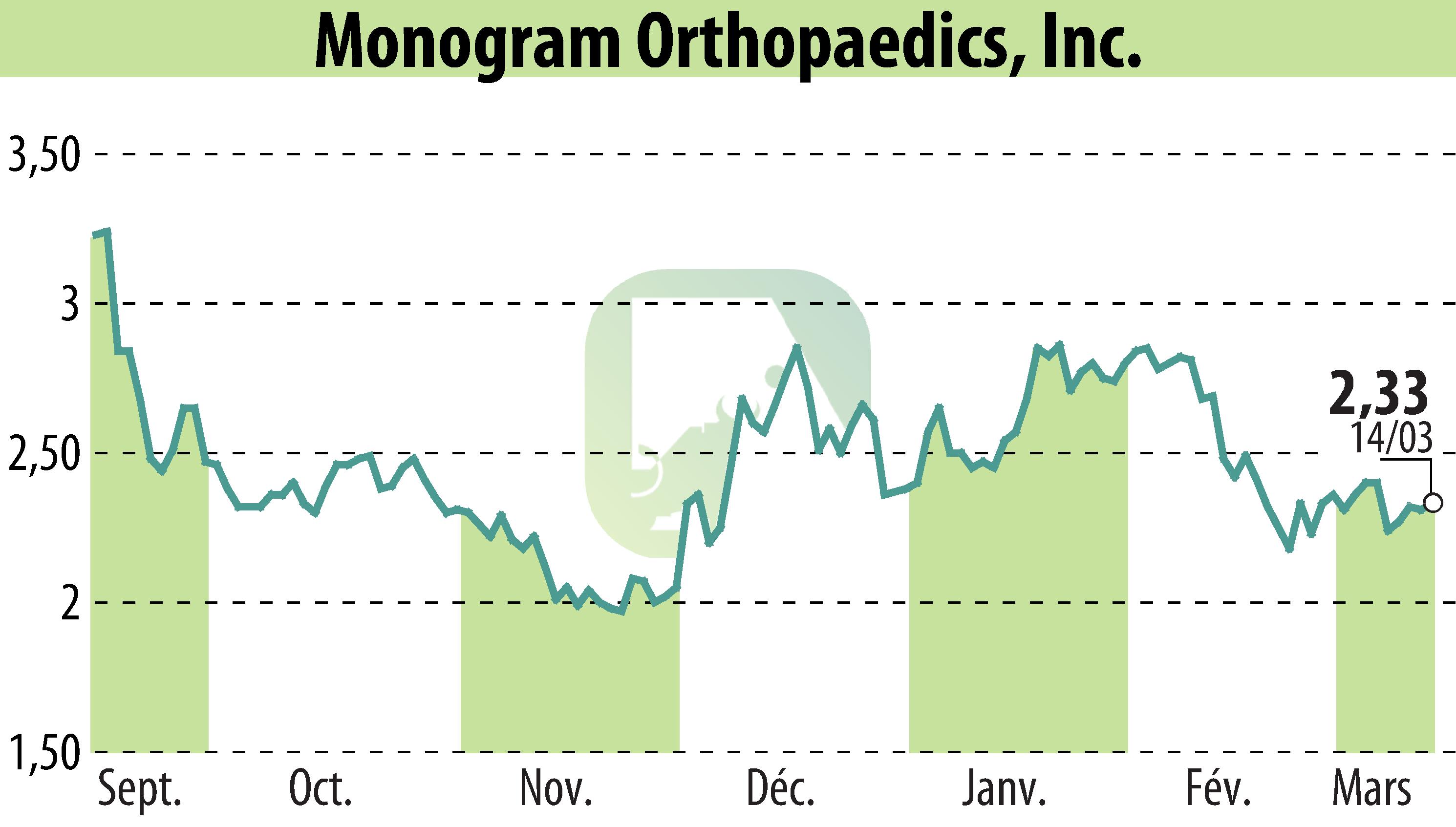
Monogram Technologies Inc. has received FDA 510(k) clearance for its Monogram mBôs™ TKA System. This clearance marks a significant milestone for the AI-driven robotics company, enabling the commercialization of its robotic-assisted technology for orthopedic surgery. The Monogram mBôs TKA System is developed to enhance safety, efficiency, and accuracy in total knee arthroplasty. The system is designed with scalability to support multiple orthopedic applications.
Ben Sexson, CEO of Monogram, emphasized the team's dedication in achieving this milestone and the potential for market impact. The company is focused on refining its strategy to support broader adoption and plans initial placements with key surgeons in strategic areas. Despite a highly consolidated market, the company aims to leverage its innovative technology for growth.
Monogram plans to integrate upgrades and system enhancements to improve competitiveness. The company is also exploring international opportunities, including potential clinical trials in collaboration with Shalby Limited in India. Its long-term strategy emphasizes gradual market adoption and technological refinement to expand the system's capabilities.
R. P.
Copyright © 2025 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all MONOGRAM ORTHOPAEDICS INC news
